RNA Trans-splicing Comes of Age
Trans-splicing is an exciting approach that RSRT is pushing forward as part of our Roadmap to Cures. I wanted to describe this promising approach to our community using a fun and accessible analogy - making a movie - that seems especially apropos given the recent Academy Awards. I hope you enjoy it and that it gives you a clear sense of how trans-splicing works. I look forward to keeping you updated in the months ahead.
A quick introduction to RNA splicing
In biology, information generally flows from DNA → RNA → protein. DNA stores the instructions needed to build and maintain our cells. Individual genes are like files on a computer, each containing specific information. When a cell needs to make a protein, it first copies the information from the gene into RNA. This RNA contains the instructions for building the protein, but also extra material that needs to be removed. The process of editing out these extra pieces is called RNA splicing. A helpful analogy is the process of making movies. DNA is like the original screenplay, and filming is like the cell generating the RNA. When you film a movie, the raw footage contains all the necessary scenes and extras like bloopers and scenes that are edited for time. RNA splicing is the editing process, removing the extras before producing the final movie (the protein).
What is trans-splicing?
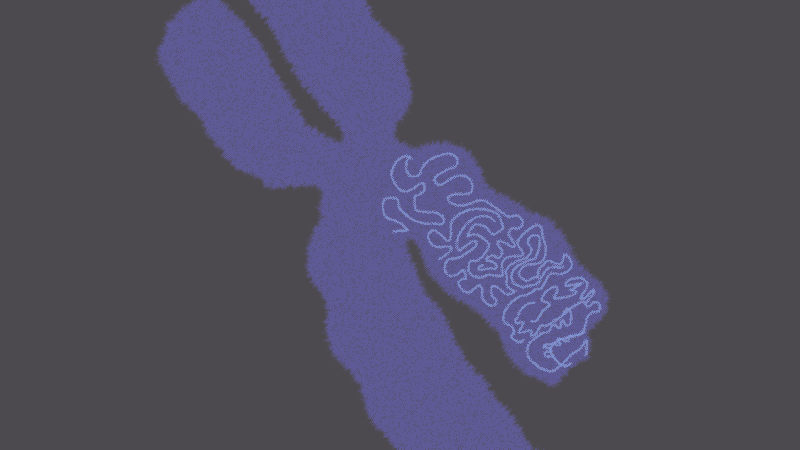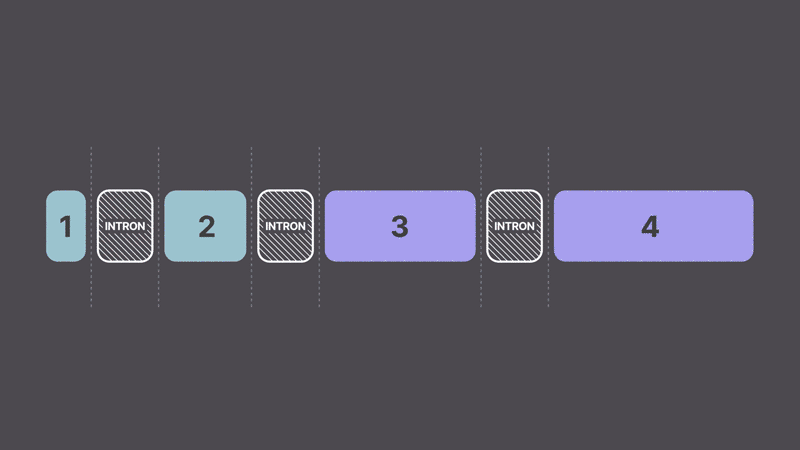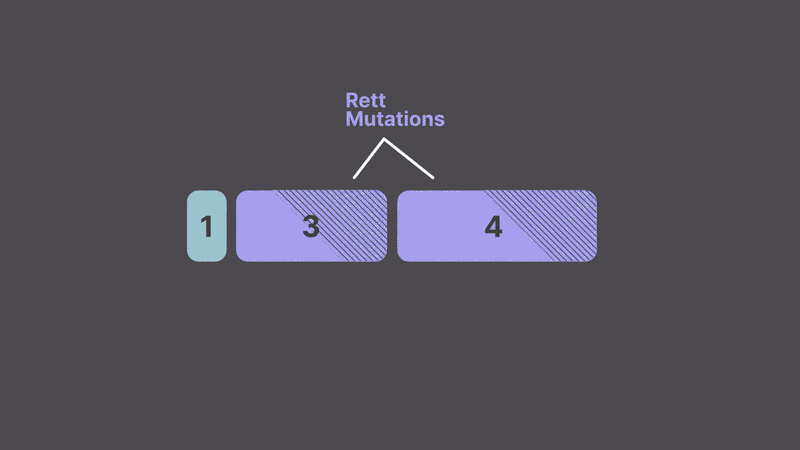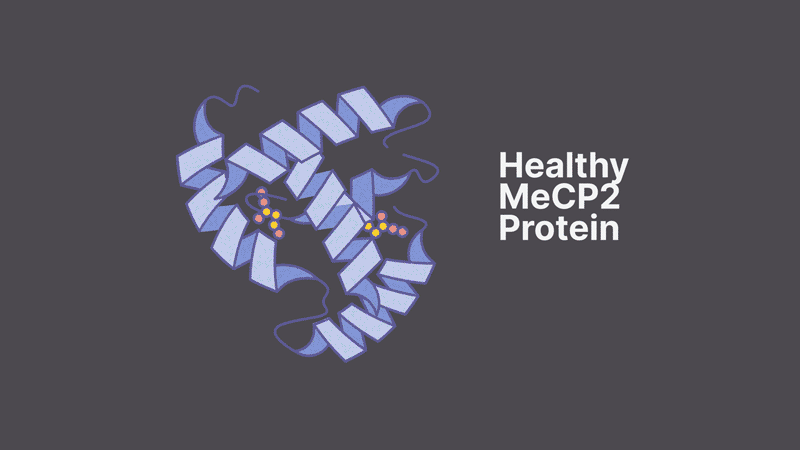
Now imagine that after filming is complete, you realize the original script contains a scene that doesn’t make sense (a mutation) and ruins the story. You could rewrite the script (DNA editing), but then you would have to re-film the movie. Alternatively, you could film a sequel that explains the mistake (gene therapy). But what if you could swap in a scene from another movie that does make sense when you are removing all the extras? That is exactly what trans-splicing does. Instead of correcting the DNA or adding a new copy of the gene, trans-splicing replaces the mutated region of RNA, allowing the cell to make the correct protein.
Why this matters for Rett Syndrome
The mutations that cause Rett Syndrome are clustered together in regions that can be replaced with a corrected version using trans-splicing molecules that take advantage of the cell’s splicing machinery. This approach offers several key advantages:
- The replacement RNA is delivered by adeno-associated virus (AAV) and can be paired with next-generation AAV delivery systems, such as the capsid developed by Apertura.
- By correcting the existing MECP2 RNA instead of adding additional copies of the MECP2 gene, there is a lower risk of potentially harmful over-expression.
- One trans-splicing therapeutic would address 97% of all Rett mutations and a second therapeutic would address the remaining 3%.
So, what’s the catch? Although trans-splicing was first demonstrated to correct genetic mutations in 1999, for many years, it simply wasn’t effective enough to be used as a genetic medicine. That has changed with advances in technology and a deeper understanding of RNA biology. Recently, several trans-splicing therapies have entered clinical trials for eye diseases and brain cancer.
RSRT’s longstanding interest in trans-splicing
Early on, RSRT recognized the potential of trans-splicing for Rett syndrome. As part of the Gene Therapy Consortium, Stuart Cobb, from the University of Edinburgh, explored this strategy as a way to maintain safe MECP2 levels. Like many researchers at the time, he found that editing efficiency was too low and instead shifted focus to his newly engineered self-regulating gene therapy, which ultimately became the foundation for Neurogene’s Rett syndrome program where Stuart is now the Chief Scientific Officer.
New Momentum in 2025
As the field matured and trans-splicing therapies began moving into the clinic, we saw renewed promise for applying this approach to Rett syndrome. In 2024 we made an award to the Abudayyeh-Gootenberg lab at Harvard Medical School to apply their newly discovered technology to MECP2 trans-splicing. In keeping with our philosophy of pursuing multiple shots on goal, in 2025 awards were made to two additional projects: an early proof-of-concept study with ReSplice Therapeutics and a large collaboration with Tacit Therapeutics.
Our partnership with Tacit Therapeutics
The collaboration began when David Nelles, CEO of Tacit, was a speaker at our Rett Syndrome Genetic Medicines Summit in Boston this past September. We, along with the audience of experts and families, were impressed by the compelling data he shared of RNA correction in Huntington’s Disease mouse models. A dialogue that began at the summit continued, and RSRT has now made a milestone-driven $1 million award to apply Tacit's unique approach to repair Rett syndrome mutations.
The goal of this award is to use Tacit's AI-assisted screening pipeline to design and test the ability of tens of thousands of potential donor molecules to correct MECP2 RNA in human cells. This process will take 4-6 months to complete, and top candidates from the screen will be optimized and then applied to Rett-patient-derived cells. Ultimately, these leads will be combined with next-generation delivery approaches and tested in a new RSRT-funded mouse model containing a fully humanized MECP2 gene.


